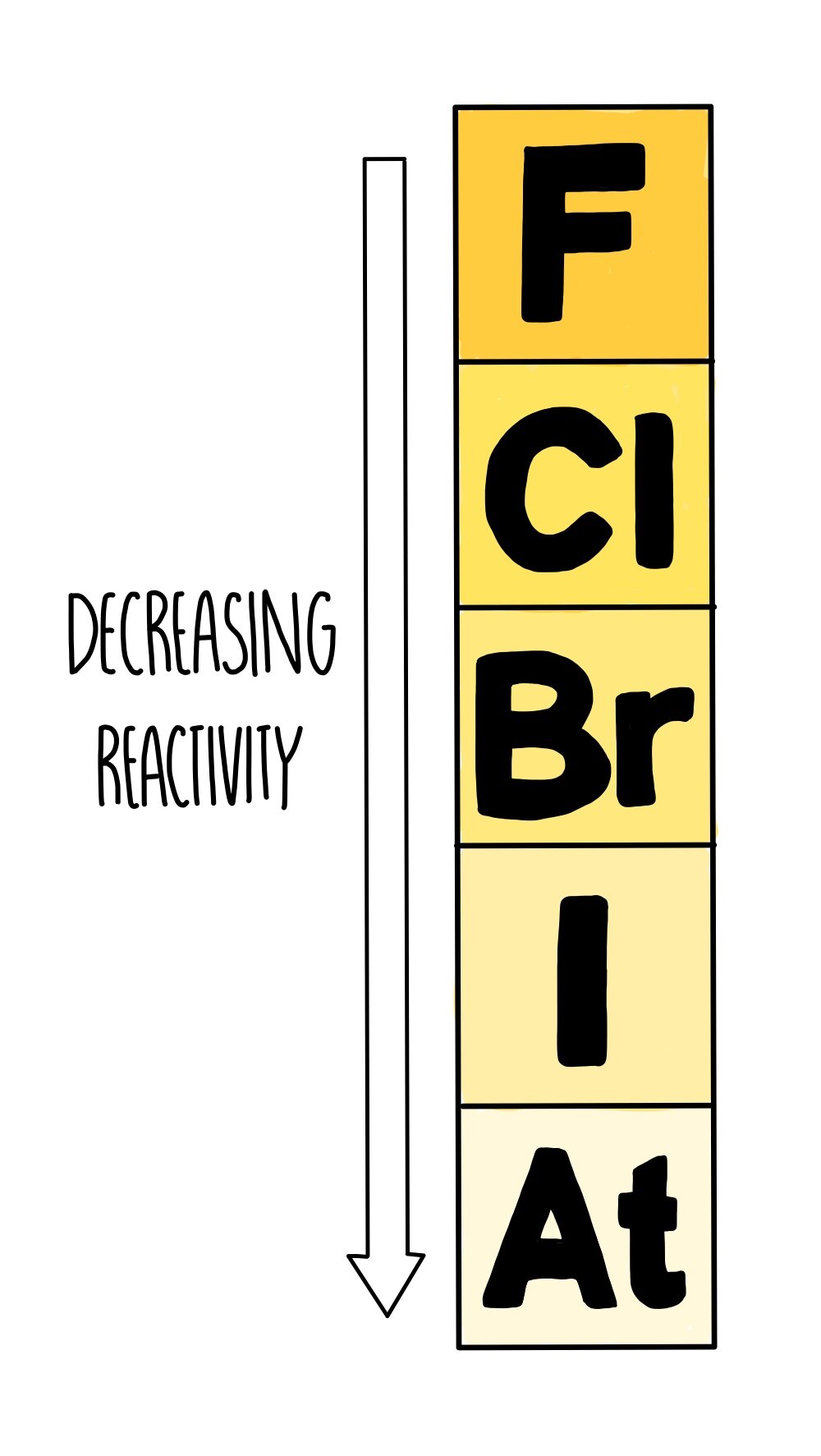
Predict the melting and boiling points of astatine, and its state at room temperature. Astatine is placed below iodine in group 7. The graph shows the melting and boiling points of the first four group 7 elements. more energy is needed to overcome these forces.the intermolecular forces become stronger.In group 7, the further down the group an element is, the higher its melting point and boiling point. The table shows the colour and physical states of chlorine, bromine and iodine at room temperature. In all groups of the periodic table, the further down the group an element is, the higher its relative molecular mass. Elements become darker in colour going down group 17 from top to bottom. Melting point and boiling point increase down Group 17 from top to bottom. First ionization energy decreases down group 17 from top to bottom. Period 2 halogen: F 2.7 + e > F 2.8 Period 3 halogen: Cl 2.8.7 + e > Cl 2.8.8 Period 4 halogen: Br 2.8.18.7 + e > Br 2.8.18.8 Period 5 halogen: I 2.8.18.18. Chemical reactivity of group 17 elements decreases down group 17 from top to bottom. Each molecule is made up of a pair of halogen atoms joined by a single covalent bond. Explaining the Reactivity Trend of the Group 7 Halogen. The halogens show trends in their physical and chemical properties. Fluorine is the most reactive element of all in Group 7. This is the opposite trend to that seen in the alkali metals in Group 1 of the periodic table. Group 7 is on the right-hand side of the periodic table, next to group 0 The non-metal elements in Group 7 known as the halogens get less reactive as you go down the group. Metal reactivity relates to ability to lose electrons. Four major factors affect reactivity of metals: nuclear charge, atomic radius, shielding effect and sublevel arrangement (of electrons). The elements in group 7 are called the halogens. The elements within the same group of the periodic table tend to exhibit similar physical and chemical properties. If you have questions about the archived correspondence, please use the VRT noticeboard.Group 7 contains non-metal elements placed in a vertical column on the right of the periodic table. The correspondence is available to trusted volunteers as ticket #2007111210016632. This correspondence has been reviewed by a Volunteer Response Team (VRT) member and stored in our permission archive. The Wikimedia Foundation has received an e-mail confirming that the copyright holder has approved publication under the terms mentioned on this page. 
If you wish to use this content, you do not need to request permission as long as you follow any licensing requirements mentioned on this page. This work is free and may be used by anyone for any purpose. As you have seen above, chlorine can also oxidise iodide ions (in, for example. 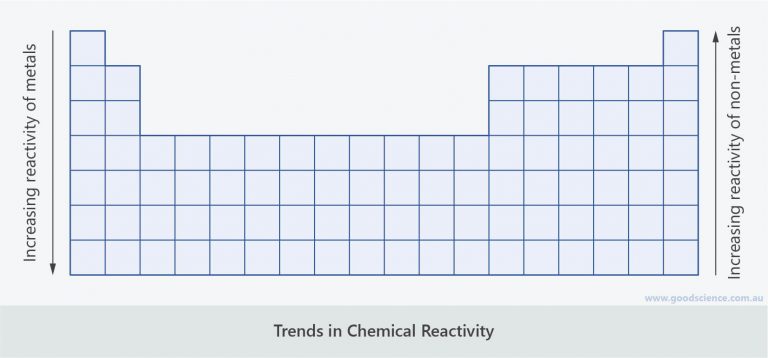 
C4 Predicting and identifying reactions and products. Non-metal atoms gain electrons when they react with metals. The bromine appears as an orange solution. C2.2.9 describe experiments to identify the reactivity pattern of Group 1 elements OCR Combined science A: Gateway. Halogen have very high electronegativities They have seven valence electrons (one short of a stable octet) They are highly reactive, therefore toxics The. The reactivity of Group 7 elements decreases down the group. For example, chlorine can oxidise the bromide ions (in, for example, potassium bromide solution) to bromine: Cl 2 + 2Br - 2Cl - + Br 2. CC BY-SA 2.5 Creative Commons Attribution-Share Alike 2.5 true true In each case, a halogen higher in the Group can oxidise the ions of one lower down.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
